When the word gram replaces mole, you have a related set of problems which requires one more step. So keep in mind that there are 4 example problems just above. Calculate the number of molecules in 1.058 mole (or gram) of H 2 O Calculate the number of atoms in 0.750 mole (or gram) of Fe These problems use the reverse technique of the above. Jan 13, 2008 First of all, you have to convert grams to number of moles by dividing the grams with its molar mass. After you get the molar mass, you have to understand that one mole contain 6.022x10^23 atoms. For chemistry you often need to convert moles to grams and grams to moles. There is a simple relation between these two:, where - mass of the substance in grams - quantity of the substance in moles - molar mass of the substance in grams/mole. And the most difficult task here is finding out the molar mass of the substance. One way to do it, assuming you know the molecular weight and how many copies of the molecules you have is as follows: 1. You calculate the molecular weight of the molecule. For example carbon dioxide, CO2, weighs 44.01 g / mol. You figure out h.
- Atoms Moles Grams Conversion Calculator
- Moles To Atoms Calculator
- Atoms To Grams Conversion Calculator Pounds
- G To Atoms Calculator
››Convert mole to atom
Please enable Javascriptto use the unit converter
››More information from the unit converter
How many mole in 1 atom?The answer is 1.660538863127E-24.
We assume you are converting between mole and atom.
You can view more details on each measurement unit:
mole oratom
The SI base unit for amount of substance is the mole.
1 mole is equal to 6.0221415E+23 atom.
Note that rounding errors may occur, so always check the results.
Use this page to learn how to convert between moles and atoms.
Type in your own numbers in the form to convert the units!
We assume you are converting between mole and atom.
You can view more details on each measurement unit:
mole oratom
The SI base unit for amount of substance is the mole.
1 mole is equal to 6.0221415E+23 atom.
Note that rounding errors may occur, so always check the results.
Use this page to learn how to convert between moles and atoms.
Type in your own numbers in the form to convert the units!
››Quick conversion chart of mole to atom

1 mole to atom = 6.0221415E+23 atom
2 mole to atom = 1.2044283E+24 atom
3 mole to atom = 1.80664245E+24 atom
4 mole to atom = 2.4088566E+24 atom
5 mole to atom = 3.01107075E+24 atom
6 mole to atom = 3.6132849E+24 atom
7 mole to atom = 4.21549905E+24 atom
8 mole to atom = 4.8177132E+24 atom
Atoms Moles Grams Conversion Calculator
9 mole to atom = 5.41992735E+24 atom
10 mole to atom = 6.0221415E+24 atom
››Want other units?
You can do the reverse unit conversion fromatom to mole, or enter any two units below:
››Common amount of substance conversions
Moles To Atoms Calculator
mole to kilomol
mole to molecule
mole to nanomol
mole to millimol
mole to picomol
mole to decimol
mole to centimol
mole to micromol
mole to molecule
mole to nanomol
mole to millimol
mole to picomol
mole to decimol
mole to centimol
mole to micromol
››Definition: Mole
The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; its symbol is 'mol.'
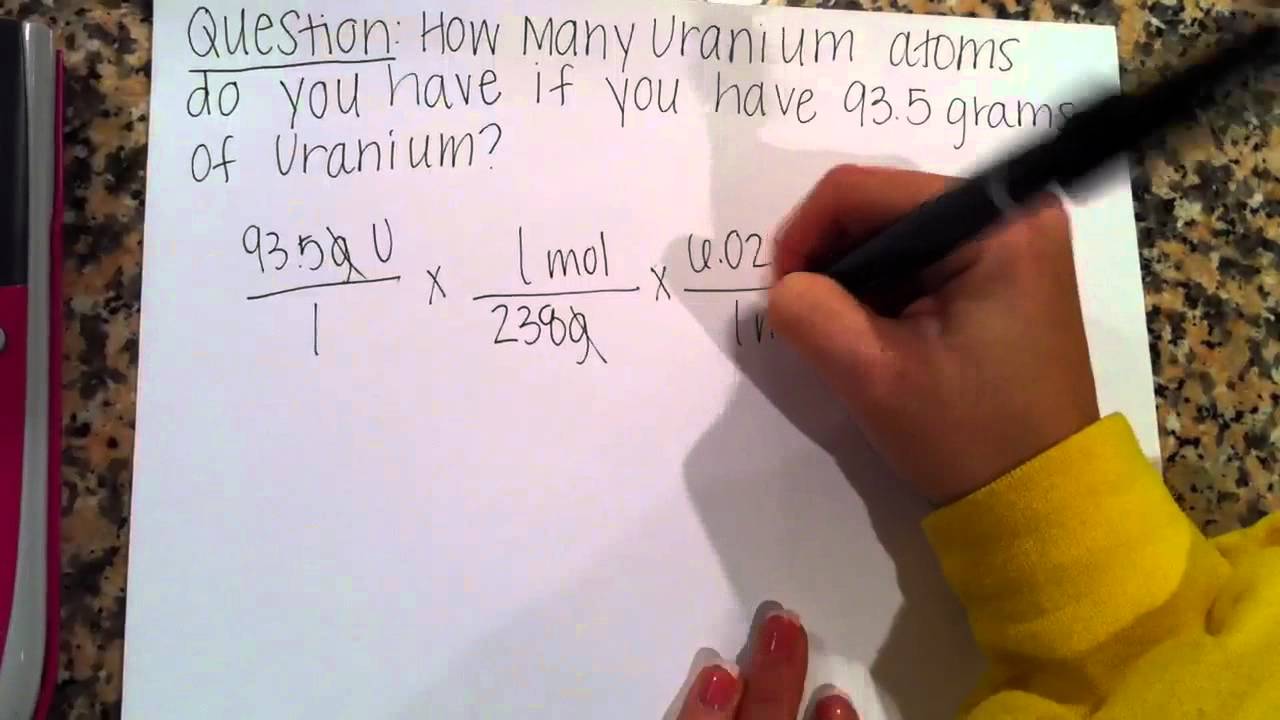
››Definition: Atom
This site uses an exact value of 6.0221415 x 1023 for Avogadro's number. This is the number of atoms in 1 mole of a chemical element.
››Metric conversions and more
Atoms To Grams Conversion Calculator Pounds
ConvertUnits.com provides an onlineconversion calculator for all types of measurement units.You can find metric conversion tables for SI units, as wellas English units, currency, and other data. Type in unitsymbols, abbreviations, or full names for units of length,area, mass, pressure, and other types. Examples include mm,inch, 100 kg, US fluid ounce, 6'3', 10 stone 4, cubic cm,metres squared, grams, moles, feet per second, and many more!
G To Atoms Calculator
•••Emilija Randjelovic/iStock/GettyImages
By Dylan Kelly
Converting atoms to grams is an essential process in basic chemistry and forms the foundation for the more difficult calculations used in more advanced chemistry. The conversion requires a fundamental understanding of Avogadro’s Number, atomic weights, dimensional analysis and the definition of a mole of a substance. Using these items, if you know how many atoms of a substance you are dealing with, you can easily convert this to grams using the following process.
Instructions
- Once you have done this a few times, you will see that multiplying the number of atoms and dividing the result by Avogadro’s number (6.02 x 10^23) will suffice.
- Use only as many significant figures as the least precise number in your calculation. In this example, 14 is two significant figures, so our answer is to two figures as well.
For the purpose of this demonstration we will assume that we are working with 14 atoms of carbon. Write “14 atoms C†on the top left of your scratch paper.
Avogadro’s number (6.02 x 10^23) is the number of particles of a substance in one mole (mol) of that substance. There are 6.02 x 10^23 atoms of carbon in a mole of carbon and 6.02 x 10^23 molecules of water in a mole of water. Because you are using dimensional analysis to cancel out “atoms,†to the right of what you wrote in Step 1 write as a fraction, “1 mol C / 6.02 x 10^23 atoms†and prepare to multiply across, such that your equation looks like this so far:
14 atoms C 1 mol x ------------------------------- 6.02 x 10^23 atoms C
Refer to the periodic table of elements and find the atomic weight for the substance you are working with, rounding to the appropriate number of significant digits. In this case, carbon has an atomic weight of 12.0 atomic mass units (amu). The molar mass (in grams) of any substance is always numerically equal to its formula weight (in amu), so for carbon, there are 12.0 grams (g) in one mole of carbon. Write this as a fraction to the right of Step 2, again multiplying. Also, put an equal sign to the far right. It should look like this:
14 atoms C 1 mol 12.0 g C x ------------------------------ x -------------- = 6.02 x 10^23 atoms C 1 mol C
Because the units in fractions get treated the same as numbers do, the “atoms C†from Steps 1 and 2 cancel each other out and the “mol C†from Steps 2 and 3 cancel out, leaving you with grams (g) as the unit of measurement your answer will be in. This is a good way to check your work.
Multiply across the top to get 168 g C and across the bottom to get 6.02 x 10^23.
Divide the top by the bottom, minding significant figures, to get 2.8 x 10^22 grams of carbon in 14 atoms of carbon.
- “Chemistry: The Central Science;†Brown, Bursten, & LeMay; 1997
